Tags
BUTLER-VOLMER Equation: Multistep Electrode Reaction, stoichiometric number, transfer coefficients
M.Sc (Chemistry)
ELECTRODE KINETICS – ELECTRODICS-II
(BUTLER-VOLMER Equation: Multistep Electrode Reaction)
Multi-step Electrode Reaction: There are several electrochemical reactions, like red-ox reaction of an organic compound on an electrode system, involving more than one electron.These reactions proceed in a multi-step way (electrochemical & non-electrochemical) involving more than one intermediate.
Electrode reactions in fuel cells; many organic red-ox reactions like the electro chemical reduction of nitro compounds etc. occur in this way.
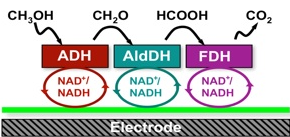 Examples of inorganic multi-step reactions:
Examples of inorganic multi-step reactions:
Reduction of Cr2O72- to Cr3+( 6 electron process), MnO4– to Mn2+( 5 electron process) ; O2 to OH–( 4 electron process) ; Fe3+ to Fe( 3 electron process).
However, even in a multi-step multi electron electrode system, only one electron will be added in an electrochemical step.
The electrode reactions can involve from simple to complex & even complicated. Hence, one can think of different kind of Butler-Volmer equation depending on the nature of the electrode reaction.
Multi-step electrode reaction can be mainly divided into TWO types depending on the nature of rate determining step (RDS)
MULTISTEP ELECTRODE REACTION-SIMPLE
The multi-step multi-electron reaction occurs in more number of steps such that each one is an elementary step (Electrochemical; Non-Electrochemical) including the rate determining step (RDS) occurring as an elementary step like any other step of the reaction.
The reaction scheme, the related discussion & derivation are given in the file in LINK-1
LINK-1: 05-BV Multi Step (Simple)
MULTISTEP ELECTRODE REACTION-COMPLEX
The multi-step multi-electron Reaction is termed complex in the sense the rate determining step (RDS) itself can be a multi-step reaction occurring in υ number of times, known as stoichiometric number.
The reaction scheme differs only in the RDS step
The reaction scheme, the related discussion & derivation are given in the file in LINK-2
LINK-2: 06-BV Multi Step(Complex)
&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&
Please go to CONTENTS for all the Articles in PoC: Click below
https://dradchem.wordpress.com/2015/06/22/properties-of-chemistry-poc-contents-2/
