Tags
Associated Legendre Functions, Microwave and far-infrared spectra, rigid rotor, ROTATIONAL SPECTRUM
The rigid rotor is a mechanical model that is used to explain rotating systems. An arbitrary rigid rotor is a three-dimensional rigid object, such as a top. A special rigid rotor is the linear rotor which requires only two angles to describe its orientation. An example of a linear rotor is a diatomic molecule. More general molecules like water (asymmetric rotor), ammonia (symmetric rotor), or methane (spherical rotor) are typical three-dimensional rotors.
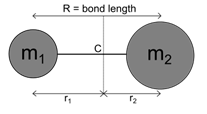 The linear rigid rotor model consists of two point masses located at fixed distances from their center of mass. The fixed distance between the two masses and the values of the masses are the only characteristics of the rigid model.
The linear rigid rotor model consists of two point masses located at fixed distances from their center of mass. The fixed distance between the two masses and the values of the masses are the only characteristics of the rigid model.
Quantum mechanical linear rigid rotor
The linear rigid rotor model can be used in quantum mechanics to predict the rotational energy of a diatomic molecule. The rotational energy depends on the moment of inertia for the system,I .
In the center of mass reference frame, the moment of inertia is
I = μr2
Where; μ is the reduced mass of the molecule and R = (r1+r2), is the distance between the two atoms as pictured above. According to quantum mechanics, the energy levels of a system can be determined by solving the Schrodinger equation:
HΨ=EΨ
To solve the Schrödinger equation for the rigid rotor, we will separate the variables and form single-variable equations that can be solved independently. Only two variables θ and φ are required in the rigid rotor model because the bond length, r, is taken to be constant. We first write the rigid rotor wave functions as the product of a theta-function, Θ depending only on θ and a phi-function, Φ depending only on φ
Ψ(θ,φ)=Θ(θ)Φ(φ)
We then substitute the product wave function and the Hamiltonian written in spherical coordinates, into the Schrödinger Equation.
HΨ(θ,φ) =EΨ((θ,φ)
Finding the Θ and θ functions is more complicated process involving separation of the variables (θ,φ) into two different equations, which on solving gives the two functions. The solution for the Θ function has been found to be
Θm(φ)=(2π)− ½ exp(±imφ) ; with m=0, ±1, ±2,⋯
The solutions to θ functions are found to be a set of power series called Associated Legendre Functions, which are power series of trigonometric functions, i.e. products and powers of sine and cosine functions.
The detailed solving and the solution for Θ and θ functions, along with their normalization constants, are shown in the following LINK-1.
LINK-1:QC-4.1 Θ and θ functions
Application of the concept of Rigid Rotor
ROTATIONAL SPECTRUM (Microwave and far-infrared spectra)
For a linear molecule, analysis of the rotational spectrum provides values for the rotational constant and the moment of inertia of the molecule, and, knowing the atomic masses, can be used to determine the bond length directly. For diatomic molecules this process is straightforward.
Rotational spectra consist of multiple lines spaced nearly equally apart because many rotational levels are populated at room temperature and the rotational energy level spacing increases by approximately 2B with each increase in J. The rotational constant, B, can be used to calculate the bond length of a diatomic molecule.
For linear molecules with more than two atoms it is necessary to measure the spectra of two or more isotopologues, such as 16O12C32S and 16O12C34S. This allows a set of simultaneous equations to be set up and solved for the bond lengths). It should be noted that a bond length obtained in this way is slightly different from the equilibrium bond length. This is because there is zero-point energy in the vibrational ground state, to which the rotational states refer, whereas the equilibrium bond length is at the minimum in the potential energy curve.
Transitions between rotational states can be observed in molecules with a permanent electric dipole moment. A consequence of this rule is that no microwave spectrum can be observed for centro symmetric linear molecules such as N2 or HCΞCH, which are non-polar.
Tetrahedral molecules such as CH4, which have both a zero dipole moment and isotropic polarizability, would not have a pure rotation spectrum but for the effect of centrifugal distortion; when the molecule rotates about a 3-fold symmetry axis a small dipole moment is created, allowing a weak rotation spectrum to be observed by microwave spectroscopy
&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&
Please go to CONTENTS for all the Articles in PoC: Click below
https://dradchem.wordpress.com/2015/06/22/properties-of-chemistry-poc-contents-2/
